A research team led by Sun Yat-sen University Cancer Center (SYSUCC) has reported a landmark advancement in precision oncology. The study, titled "Risk-adaptive Therapy Guided by Dynamic ctDNA in Nasopharyngeal Carcinoma," was published in the world-leading journal Nature on March 12, 2026.
The research was co-led by Prof. Ying Sun and Associate Prof. Jiawei Lv from SYSUCC. This milestone represents another breakthrough by the team in the field of precision diagnosis and treatment for nasopharyngeal carcinoma (NPC).

For decades, cancer treatment has largely followed a fixed-course approach—patients receive predetermined therapies based on guidelines established before treatment begins. However, tumor cells and their microenvironments continuously evolve in response to treatment, meaning that a patient's risk of recurrence and metastasis changes dynamically throughout therapy. This fundamental mismatch has long troubled clinicians: some low-risk patients may be overtreated and suffer unnecessary toxicities, while high-risk patients may receive insufficient treatment intensity, missing the critical window for intervention.
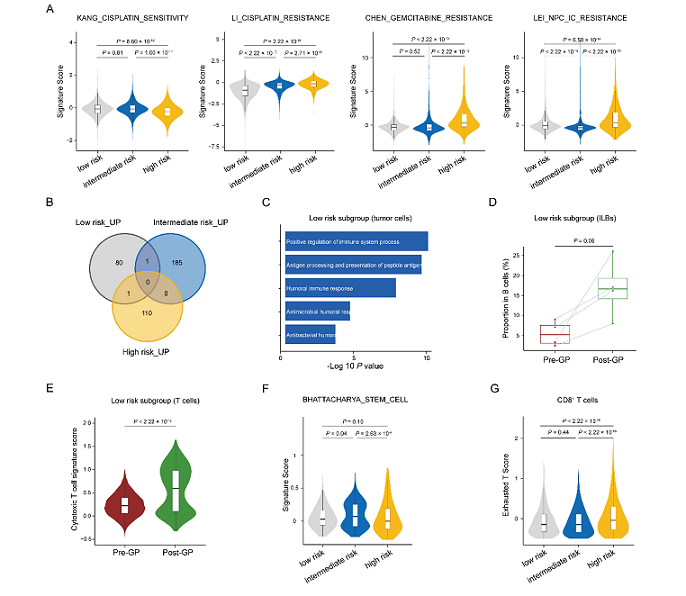
The team pioneered a solution by identifying a non-invasive molecular marker capable of reflecting patients' real-time risk profiles. Building on their 2024 Cancer Cell publication (the "EP-SEASON" study), which established that dynamic changes in plasma Epstein-Barr virus DNA (cfEBV DNA) during treatment serve as a reliable biomarker for NPC treatment response and recurrence risk, the researchers designed and conducted the "EP-STAR" trial.
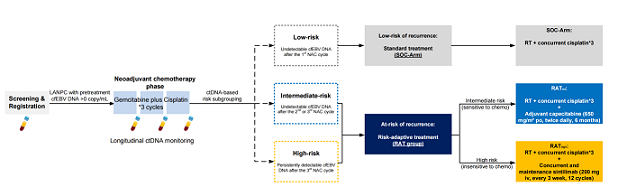
This multicenter, phase II adaptive clinical trial leveraged the "window of opportunity" during induction chemotherapy to monitor patients' cfEBV DNA clearance patterns. Based on these dynamics, patients were stratified into three risk groups, with subsequent treatment regimens adapted accordingly:
• High-risk group: cfEBV DNA remained detectable after three cycles—received risk-adaptive therapy (RAThigh) with anti-PD-1 immunotherapy (sintilimab) starting from radiotherapy initiation and continuing for 12 maintenance cycles.
After a median follow-up of 47.3 months, the RAT strategy demonstrated remarkable efficacy. The 3-year failure-free survival rate reached 89.1% (95% CI: 83.2–95.0%) in the RAT group: 91.4% in the intermediate-risk group and 86.5% in the traditionally poor-prognosis high-risk group. The low-risk group receiving standard treatment achieved a 90.6% 3-year failure-free survival rate.
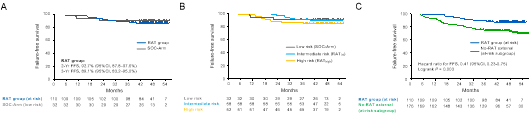
Notably, compared with patients from the prospective EP-SEASON cohort who had identical risk profiles but did not receive adaptive therapy, the RAT strategy significantly reduced the risk of disease progression or death by 59% (hazard ratio = 0.41, 95% CI = 0.23–0.75, P = 0.004).
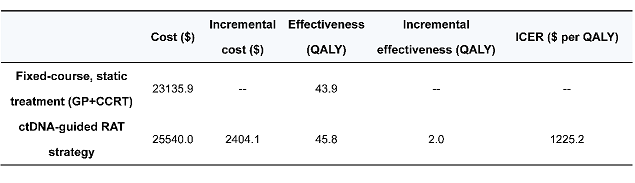
The strategy demonstrated favorable safety profiles without compromising quality of life. Markov model analysis revealed that although the RAT strategy increased total costs by $2,404.1, it gained 1.9 quality-adjusted life years (QALYs), with an incremental cost-effectiveness ratio of $1,225.2 per QALY—well below the willingness-to-pay threshold, confirming its cost-effectiveness.
In an accompanying commentary, Nature Associate Editor Dr. Victoria Aranda noted that the study provides valuable new insights into ctDNA and personalized medicine, extending beyond EBV-associated NPC to offer landmark guidance for future clinical trial design and clinical practice transformation.

Co-first authors: Associate Prof. Jiawei Lv, Postdoctoral Fellow Danxue Zheng (SYSUCC), Prof. Jinhui Liang (Wuzhou Red Cross Hospital), Prof. Ning Zhang (Foshan First People's Hospital), Senior Technician Zulu Ye, Postdoctoral Fellow Xudong Xu (SYSUCC), and Prof. Melvin L.K. Chua (National Cancer Centre Singapore).
This work was supported by the National Natural Science Foundation of China Innovative Research Group Project (Category A).
Link to the original article: https://www.nature.com/articles/s41586-026-10244-w?sessionid=-516133757
Copyright:Sun Yat-sen University Cancer Center Designed by Wanhu.