
The primary endpoint of 18wPFS rate was met with a rate of 43.8% (21 of 48) for the entire study population. Secondary endpoint results include a median PFS of 3.7 months, an overall response rate of 29.2% (14 of 48), a disease control rate of 56.3% (27 of 48) and a median duration of response of 12.0 months. The secondary endpoint of median overall survival time was not mature. The triplet arm exhibited significantly improved outcomes compared to the doublet arm, with a higher 18wPFS rate (64.0% versus 21.7%) and longer median PFS (7.3 months versus 1.5 months) (Figure 1).

Figure 1
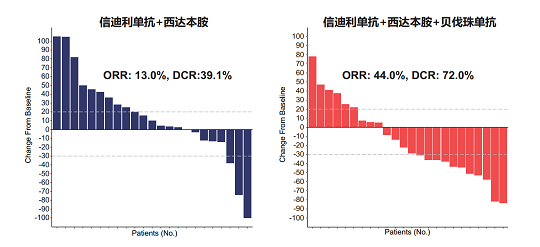
Figure 2
Specifically, among the 26 patients with liver metastasis, those in the triplet arm exhibited higher 18wPFS rate (64.3% versus 8.3%), longer median PFS (7.3 months versus 1.4 months) (Figure 3), higher ORR (50.0% versus 8.3%,) and higher DCR (71.4% versus 8.3%) compared to those in the doublet arm.
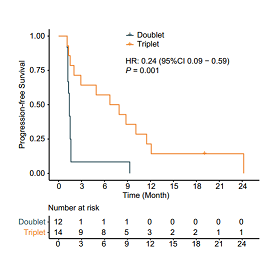
Figure 3
Analysis of bulk RNA sequencing data from the patients suggested that the triplet combination enhanced CD8+ T cell infiltration, cytotoxic lymphocytes and monocytic lineage, coupled with a concomitant decrease in intratumoral B lineage, endothelial cells and fibroblasts following treatment. Gene set variation analysis (GSVA) provided further insights into the molecular mechanisms. Among the responders in the triplet therapy group after treatment, a stronger antitumor immune response was observed across the entire process from antigen presentation to effector cell killing ability (Figure 4).
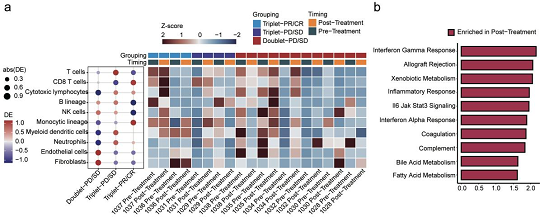
Figure 4
In order to break the dilemma of immunotherapy in colorectal cancer, the team was devoted in this field for many years, from laboratory to clinical transformation, and boldly proposed the original innovative strategy of the triplet combination. The triplet combination showed superior therapeutic efficacy than the doublet regimen, and the potential underlying mechanism of this observed efficacy involves the infiltration and the activation of CD8+ T cells.
CAPability-01 studies the novel combination of epigenetic drug + immune check point inhibitor + anti-angiogenic drug, with high potential in the treatment in more cancer types. Based on the results of CAPability-01 research, CAPability-02 has been started with larger sample size. The exploration of the triplet combination in the second line treatment of metastatic colorectal cancer has been carried out in 17 centers across China.
Professor Ruihua Xu and Professor Feng Wang from Sun Yat-sen University Cancer Center are the corresponding authors of this paper. Professor Feng Wang, Assistant Chief Physician Jin Ying, Dr. Wang Min and Chief Physician Luo Huiyan from Sun Yat-sen University Cancer Center, and Professor Weijia Fang from the First Affiliated Hospital, Zhejiang University School of Medicine are the first authors of this paper.
Link to the original article: https://www.nature.com/articles/s41591-024-02813-1
Written by: Liao Shuang, Zhai Huiwen
International Office,
Sun Yat-sen University Cancer Center
Copyright:Sun Yat-sen University Cancer Center Designed by Wanhu.